
The US-based Arcturus Therapeutics Holdings Inc. will collaborate with Vinbiocare Biotechnology Joint Stock Company, a member of Vingroup, to establish a manufacturing facility in
Under an agreement announced on August 2, Vinbiocare will build out a manufacturing facility in
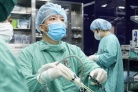
Arcturus Therapeutics’ scientists study vaccines in the laboratory. Photo: ARCTURUS |
Arcturus will allow Vinbiocare access to proprietary technologies and processes for the manufacture of Arcturus’ investigational Covid vaccines. Arcturus will provide Vinbiocare with an exclusive license to manufacture the vaccines in
“The license and technology transfer applies toward drug product manufacturing but not toward mRNA drug substance manufacturing. Technology transfer is underway,” according to a press release of the Arcturus Therapeutics Holdings Inc.
Vinbiocare will make an upfront payment of
Joseph Payne, President and CEO of Arcturus, said under the agreement the Vingroup’s subsidiary Vinbiocare, his holding would help build a manufacturing facility for Covid-19 vaccine candidates, targeting a capacity of up to 200 million doses per year.
“We are looking forward to having a meaningful role in providing effective and safe Covid-19 vaccine to
VinBioCare factory is under construction in the Hoa Lac Hi-tech Park on the outskirts of Hanoi, with an investment of over US$200 million. This is one of the five key projects having received the Hanoi authority's approval during the social distancing period in the capital city.
VinBioCare is projected to produce the first batches of vaccines by early 2022.
The country is working to speed up Covid-19 vaccination for its population of nearly 100 million. Some 5.5 million people have received one shot of the vaccine while nearly 550,000 have been completely immunized with two shots.



- Hanoi launches pilot project to integrate electronic health records into VNeID app
- Modern pediatric hospital opens in Hanoi
- Hanoi to protect children against measles, rubella
- Hanoi works on cleanup, disease prevention after Typhoon Yagi
- Snoring and mouth opening/breathing during sleep can be signs of severe sleep apnea
- Hanoi, central provinces hold events to raise awareness of drowning prevention

