
About 150 students from medical and pharmaceutical universities in Hanoi have volunteered to join in a trial for Vietnam-made vaccine of Covid-1.
Launched by Nanogen Pharmaceutical Biotechnology JSC and the Vietnam Military Medical Academy in early December, the trial has so far received many proposal from volunteers.
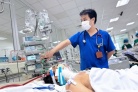
Volunteers register to participate in the clinical vaccine trial program at the Vietnam Military Medical Academy. Photo: Kinhtedothi.vn |
Before the testing, volunteers have to fill out the consent form and stay at the academy in at least 72 hours to confirm their qualified health condition.
Director of the Vietnam Military Medical Academy Do Quyet said that the volunteers of the phase 1 will be examined and screened before being injected as requested by the Ministry of Health. There are total three testing phases - all must comply with regulations to ensure absolute safety.
The volunteers will be discharged and instructed to follow up and take notes themselves at home. Every day the medical staff will visit or call them.
The Ministry of Health has been streamlining the administrative process to put Nanocovax vaccine into use at the shortest time.
“Nanogen can produce two million doses of vaccine per year. During the clinical trials, we will produce the Covid-19 vaccine while upgrading the factory to optimize capacity to some 30 million doses per year," Ho Nhan, the company’s director, said.
"We plan to reach an ideal capacity of 50 million doses per year,” he added.
"This is the first clinical trials of locally-made Covid-19 vaccine that whose research process has completed and achieved high results in the pre-clinical trial. We are making every effort to be able to produce vaccines as soon as possible," Mr. Nhan added.
Aside from Nanogen Company, three other units are developing Covid-19 vaccine in Vietnam, including Vabiotech, Polyvac and the Institute of Vaccines and Medical Biologicals (IVAC). Among them, IVAC and Vabiotech may conduct human trial in February and March 2021, respectively.
From December 10, those wishing to join Nanocovax anti-Covid-19 candidate vaccine clinical research program can register directly at the Center for Clinical Trial and Bioequivalence of the Vietnam Military Medical Academy (No. 222 Phung Hung street, Ha Dong district in Hanoi) or by phone: 0388.959.096, 0362.359.084 / 0353.212.500. Alternatively, registration can be made via email: nanocovax@nanogenpharma.com or on the website: nanogenpharma.com.



- Hanoi launches pilot project to integrate electronic health records into VNeID app
- Modern pediatric hospital opens in Hanoi
- Hanoi to protect children against measles, rubella
- Hanoi works on cleanup, disease prevention after Typhoon Yagi
- Snoring and mouth opening/breathing during sleep can be signs of severe sleep apnea
- Hanoi, central provinces hold events to raise awareness of drowning prevention

